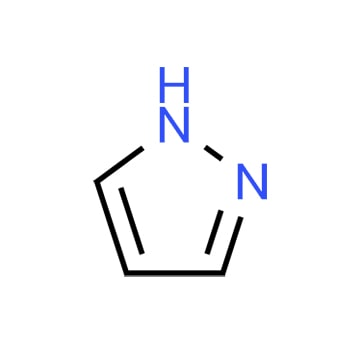
Pyrazole, also known as 1H-pyrazole or 1,2-Diazole, is an important organic compound with a variety of applications.
Pyrazole has remarkable conjugated structure and aromaticity, with two nitrogen atoms in different nitrogen atom states on its ring, which make it different from other aromatic compounds. Meanwhile, its electron withdrawing and conjugation properties also gives it good electrophilicity and acid-base properties, making it widely used in fields such as pharmaceutical chemistry, material chemistry, organic synthesis, biology, and etc.
Properties of Pyrazole
1H-pyrazole is a white to yellow crystal powder under room temperature, with good solubility in organic solvents such as ethanol, ether, chloroform, benzene, but slightly soluble in water.
With other physical properties as below:
Melting point: 67-70 °C (lit.)
Boiling point: 187.0±9.0 °C at 760 mmHg
Flash Point: 87.5±11.7 °C
Structural characteristics of Pyrazole
1H-pyrazole is a five membered nitrogen heterocycles aromatic compound. The nitrogen atom in its structure has strong nucleophilicity, which makes it able to undergo nucleophilic substitution reactions with various electrophilic alkylation reagents to obtain corresponding pyrazole derivatives.
Applications of Pyrazole
In medicine field: 1H-pyrazole is often used as a pharmaceutical intermediate to form building blocks for drug molecules. Pyrazole and its derivatives have various pharmacological activities such as antibacterial, antiviral, anti-tumor, anti-inflammatory, and antidepressant, and are therefore widely used in the pharmaceutical field to treat cancer, cardiovascular disease, dementia, diabetes, etc.
In agrochemical field: 1,2-Diazole is used for structural modification and synthesis of organic molecules with insecticidal, bactericidal, and herbicidal activities.
In organic synthesis: Pyrazole, as an aromatic five membered nitrogen heterocycle, has strong chemical conversion activity. It can easily undergo chlorination, bromination, iodization, alkylation, and acylation reactions to obtain pyrazole derivatives with diverse structures and special properties,
In biology field: 1,2-Diazole and its derivatives can serve as biological probes for detecting specific molecules and active substances in living organisms. For example, pyrazole compounds can bind to metal ions or proteins for use as fluorescent probes, colorants, etc.